|
electron-domain geometry Consider the molecule PF4Cl. Therefore, this structure is the stable Lewis structure of PF 3. A quick explanation of the molecular geometry of PF3 including a description of the PF3 bond angles. a) Draw a Lewis (electron dot) structure of phosphine. Question 3 Phosphine (IUPAC name phosphane) is a hydride of phosphorus, with the formula PI-13. Lewis (electron dot) structure Molecular geometry b) Predict with a reason, whether the molecule PF3 is polar or non-polar. And the outside atoms (fluorines) also form an octet. to deduce the molecular geometry of each species. In the above structure, you can see that the central atom (phosphorus) forms an octet. Here, both phosphorus and fluorine atoms do not have charges, so no need to mark the charges. This theory explains that the bond angle between the fluorine-phosphorus-fluorine (F-P-F) is 97°. a) Draw the Lewis (electron dot) structures of PF3 and PF4+ and use the VSEPR theory to deduce the molecular geometry of each species. The geometrical structure of the tetra-atomic Phosphorus Trifluoride (PF3) molecule is studied with the help of the Valence Shell Electron Pair Repulsion (VSEPR) theory. Use the following formula to calculate the formal charges on atoms:įormal charge = valence electrons – nonbonding electrons – ½ bonding electronsįor phosphorus atom, formal charge = 5 – 2 – ½ (6) = 0įor each fluorine atom, formal charge = 7 – 6 – ½ (2) = 0 Lewis (electron dot) structures are useful models. Lone pairs marked, and got the stable Lewis structure of PF 3 #3 Indicate formal charges on the atoms, if necessary Therefore, place phosphorus in the center and fluorines on either side. Would you expect the molecule to distort from its ideal electron-domain geometry. Since phosphorus is less electronegative than fluorine, assume that the central atom is phosphorus. molecular geometry of PF3 including a description of the PF3 bond angles. We have to place the least electronegative atom at the center. If no more electrons are available and the central one does not yet have a full octet, a lone pair on a peripheral atom can be pushed into a second (or third). So the total electron pairs = 26 ÷ 2 = 13 Total electron pairs = total valence electrons ÷ 2 And when we divide this value by two, we get the value of total electron pairs. Learn how to find: Phosphorus valence electrons and Fluorine valence electrons Valence electrons of one phosphorus atom = 5 × 1 = 5 Valence electrons of three fluorine atoms = 7 × 3 = 21Īnd the total valence electrons = 5 + 21 = 26 Since PF 3 has one phosphorus atom and three fluorine atoms, so… Hence, phosphorus has five valence electrons and fluorine has seven valence electrons.  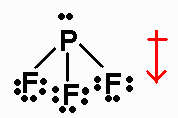
The polarity of I 3 ion is a debatable topic. The ideal electron pair geometry of I 3 is trigonal bipyramidal. The I 3 ion has a linear shape or molecular geometry. In the periodic table, phosphorus lies in group 15, and fluorine lies in group 17. The negative 1 charge present on the ion accounts for 1 extra electron added in its Lewis structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
